
Health
AISym4MED
AISym4Med aims at developing a platform that will provide healthcare data engineers, practitioners, and researchers access to a trustworthy dataset...

Project dates
2021 - 2024
Website
Role of Zabala
Partner
Project led by
HIPRA SCIENTIFIC SL (HIPRA SCIENTIF)
13
Partners
5
Countries
9
M€ budget
30
Months
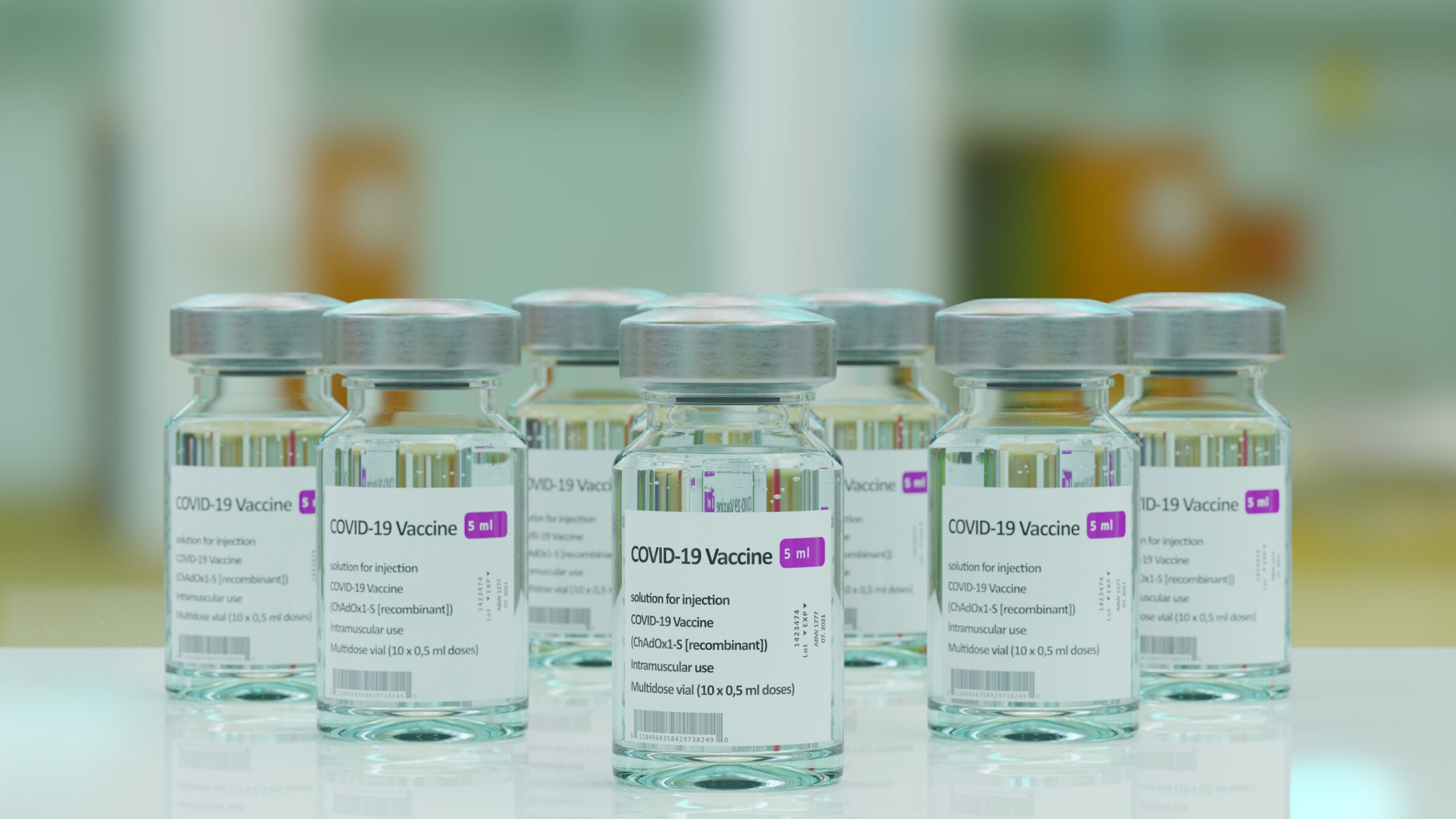
Since the publication of the genome sequence of SARS-CoV2, on January 11th, 2020, an endeavour of unprecedented speed and magnitude set out to develop a vaccine against the disease. An ideal SARS-CoV-2 vaccine should meet the following requirements: protect not only from severe disease but also thwart infection in all vaccinated populations, including less immunocompromised individuals, elicit long term memory immune responses after a minimal number of immunizations or booster doses, able to ramp up production to produce billions of doses.
RBDCOV will provide a vaccine that should meet the following requirements: protect not only from severe disease but also thwart infection in all vaccinated populations, including less immunocompromised individuals; elicit long term memory immune responses after a minimal number of immunizations of booster doses; able to ramp up production to produce billions of doses annually and be easily accessible for worldwide vaccination campaigns at an affordable cost and at limited time.
RBDCOV is an ambitious project that will offer a new tool to control the pandemic in the short-medium and long term. RBDCOV aims to manufacture and test the first recombinant protein-based vaccine to be licensed in Europe beyond the scope of the project. The final formulation of RBDCOV vaccine, the company’s experience in the use of highly innovative platforms for the manufacture of vaccines animal vaccine manufacturing and the strong consortium involved in the RBDCOV project will make it feasible to lay the foundation for this challenging goal.


Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union. Neither the European Union nor the granting authority can be held responsible for them.

“RBDCOV mission was generated, trying to obtain a safe and efficient vaccine against COVID in a short term by merging for the first time the knowledge and capacities of key actors internationally renowned from the field of animal and human vaccine research and development”
Laura Sesma
Team leader expert in European Programmes in the Health Area













Health
AISym4Med aims at developing a platform that will provide healthcare data engineers, practitioners, and researchers access to a trustworthy dataset...

Agroalimentation and bioeconomy
GO-GRASS will develop a new circular and integrated agri-food system whose main application will be in grasslands and which will...

Climate, Natural Resources and Environment
InBestSoil aims to co-create an investment framework around soil conservation and restoration by developing an economic valuation system for the...

News
Launch of the last Horizon 2020 call for proposals from the EIC Pathfinder FET Innovation LaunchPad.

Opinion
MANUFACTURE OF GOODS

Marian Pereira
Senior Consultant

Publication
FINANCIAL FRAMEWORK
Have a look to our downloadable brochure with all the information, where you can see how much is allocated to each programme.
The important thing is not to keep moving, but rather to know in which direction to go. Our 37% success rate proves that we know how to guide our clients.
Our compass is our clients; our mission is to solve their problems, guiding them so that innovation becomes their key to competitiveness.

This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Strictly Necessary Cookie should be enabled at all times so that we can save your preferences for cookie settings.
This website uses Google Analytics to collect anonymous information such as the number of visitors to the site, and the most popular pages.
Keeping this cookie enabled helps us to improve our website.
Please enable Strictly Necessary Cookies first so that we can save your preferences!
This website uses the following additional cookies:
(List the cookies that you are using on the website here.)
Please enable Strictly Necessary Cookies first so that we can save your preferences!